Media
HKU chemists develop a fast and robust assay for comprehensive evaluation of bromodomain inhibitors as preclinical drug candidates in living cells
15 Jul 2019
A research team led by Dr Xiang David Li from the Department of Chemistry at The University of Hong Kong (HKU) has developed a robust chemical proteomics platform that, for the first time, enables a live cell-based comprehensive evaluation of bromodomain inhibitors, a new class of drug candidates for the treatment of a variety of severe human diseases such as cancer and diabetes. The research findings were recently published in a top chemistry journal — Journal of the American Chemical Society.
Bromodomains are a type of structure modules that exist in many different proteins. In human cells, the bromodomains play important roles in many processes that regulate the functions of our genome. Errors in the processes governed by bromodomains can cause mistakes in gene expression, leading to the development of human diseases, such as cancer.
Bromodomains have emerged as “hot spots” for drug discovery. It has attracted inputs from not only academia but also dozens of pharmaceutical giants. Multiple inhibitors are currently in different stages of clinical trials. Most of the trials have been focusing on the treatment of different types of cancers. For example, in 2012, GlaxoSmithKline started the Phase I and II clinical investigation of compound GSK525762 toward NUT midline carcinoma, non-small cell lung cancer, colorectal cancer, prostate cancer, breast cancer, and other solid tumor subjects. Two years later, the same company initiated the clinical trial for the same compound in subjects with relapsed and refractory hematologic malignancies.
Investigations on the clinical potentials of bromodomain inhibitors as therapeutic agents for other diseases have also been reported. For example, Resverlogix, a Canadian biotech company, developed a bromodomain inhibitor Apabetalone, which showed statistically significant decrease in major adverse cardiovascular events from patients with type II diabetes mellitus in Phase II trial. Early in this June, Resverlogix announced that the Phase III trial of Apabetalone has been almost completed and would report the top-line results in this September.
Aside from these successful examples, several failures or terminations in clinical trials were also reported because of unexpected side effects or toxicity caused by the tested inhibitors. Therefore, a comprehensive understanding of a drug candidate’s cellular selectivity in the early stage of drug development is important to identify potential off-targets, which should largely avoid failures in the later clinical trials. This is particularly important for developing drugs to target bromodomains, as in human, there are 61 different bromodomains that share high structural similarity. A non-selective inhibitor that targets multiple bromodomains may result in off-target side effects. Therefore, a key step in the inhibitor development is to evaluate the selectivity of developed inhibitors toward different bromodomains
However, most of the currently available methods to evaluate the selectivity of bromodomain inhibitors only work in test tubes, an environment that is significantly different from that in cells. And these methods usually use “man-made” bromodomains but not the natural ones. As a result, the selectivity evaluation may not fully reflect the real situation inside the living cells. A method that enables the evaluation of the selectivity of bromodomain inhibitors toward natural bromodomains in living cells remained lacking.
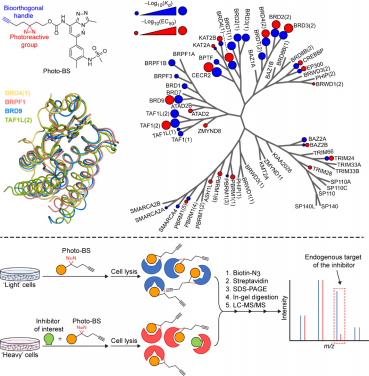
To fill this technology gap, Dr. Li’s team developed a novel chemical proteomics platform for bromodomain inhibitor selectivity evaluation in living cells. The platform relies on the combination of a newly developed chemical probe named photo-bromosporine (photo-BS) and the state-of-the-art quantitative proteomics technology. Briefly, photo-BS can bind to a variety of natural bromodomains in living cells. When being activated by UV light, photo-BS covalently captures the bromodomains. Further isolation and analysis of the captured bromodomains by mass spectrometry give the identities of them. When a bromodomain inhibitor is added into the living cells, the inhibitor will bind to its targeted bromodomain(s), occupying the photo-BS-binding site. In this way, the inhibitor-binding bromodomain(s) can no longer be captured by photo-BS, and thus disappear from the following analysis. By comparing the results obtained in the absence and presence of the bromodomain inhibitor, the bromodomain(s) that disappear(s) after the addition of the inhibitor can be identified as the selective target(s) of this inhibitor.
In the current study, Dr. Li’s team tested the performance of this platform using multiple known bromodomain inhibitors. The results matched perfectly with the reported selectivity of these inhibitors. Since more and more efforts have been made to target the bromodomains for disease treatments, and the list of the archived bromodomain inhibitors are rapidly increasing, Dr. Li’s study provides a powerful platform to evaluate the selectivity of the newly developed inhibitors at the early stage of drug discovery. Such evaluation may help avoid economic and time losses at both the industry and the patient sides caused by a non-selective inhibitor in later clinical trials. Dr. Li envisions that this platform can facilitate the further development of novel bromodomain inhibitors as promising therapeutic agents.
For more details, please see: https://pubs.acs.org/doi/10.1021/jacs.9b02738
Media enquiries:
For media enquiries, please contact Ms Cindy Chan, Assistant Director of Communications of HKU Faculty of Science (tel: (852) 3917-5286/ (852) 6703-0212; email: cindycst@hku.hk).
Image download:
https://www.scifac.hku.hk/press